
Standardise safety data collection with Reportum
Understanding patient safety is an essential element of Patient Support Programs. However, traditional safety reporting systems involve multi-step manual processes that often result in compliance

Understanding patient safety is an essential element of Patient Support Programs. However, traditional safety reporting systems involve multi-step manual processes that often result in compliance

Safety reporting is not a core focus for field-based staff, therefore inputting of reports is often completed at a later time. This means report entry

A complete picture of drug safety cannot be created if only part of the available data is captured, however the current adverse event capture process

Through the input of industry leader members of the PV Insights Network, Qinecsa focuses on providing solutions to the safety challenges facing small pharmaceutical and

A top 10 pharma company faced challenges in standardizing safety surveillance across Pharma, Vaccine, and Consumer Health units, including consolidating signal teams, facilitating a divestment,
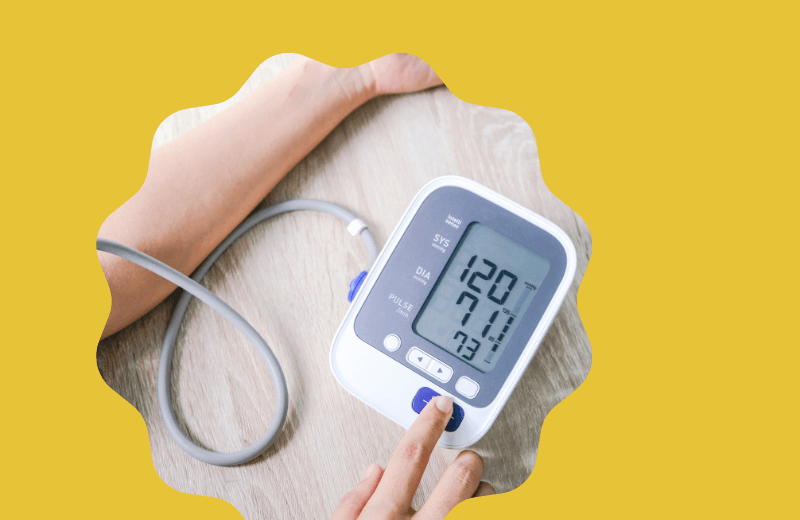
In her latest article for IPI, Qinecsa President, Humaira Qureshi discussed the practical challenges of integrating medical devices into pharmacovigilance portfolios, the obligations of device
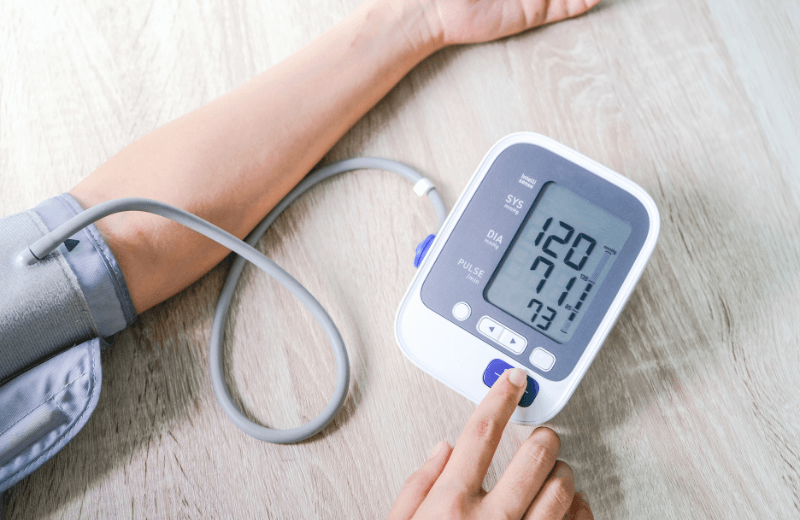
In her latest article for IPI, Qinecsa President, Humaira Qureshi, addresses the practical challenges of integrating medical devices into pharmacovigilance portfolios. She explores the obligations

A head of our first exhibition at the DIA Global Annual Meeting, taking place in San Diego from June 16th-20th, Qinecsa spoke with Outsourcing-Pharma about our

Following her attendance at DIA Europe 2024, Humaira Qureshi, President of Qinecsa Solutions, spoke with PharmaPhorum about the transforming pharmacovigilance landscape, the potential barriers and

We are thrilled to announce that Humaira Qureshi, President of Qinecsa, will be a keynote speaker at the upcoming Insife Annual Convention 2024. The event