
Building the Foundations for Proactive Drug Safety
Qinecsa Chief Product Officer, Beena Wood, has been featured in Pharm Tech’s latest issue, where she outlines how proactive PV, translational safety, AI foundations, and

Qinecsa Chief Product Officer, Beena Wood, has been featured in Pharm Tech’s latest issue, where she outlines how proactive PV, translational safety, AI foundations, and

Qinecsa Chief Product Officer, Beena Wood, has been featured in Pharm Tech’s latest issue, where she explains how AI could revolutionize pharmacovigilance, if data, trust,

Qinecsa Chief Product Officer, Beena Wood, has been featured in Pharm Tech’s latest issue, where she saw 2025 as an AI superposition in which pharmacovigilence

Qinecsa Chief Operating Officer, John Cogan, has been featured in The Pharma Navigator latest issue, where he discusses trends from 2025 for the bio/pharma industry.

Qinecsa VP, Product Management Reportum, Daniel O’Keeffe, has been featured in PharmaTimes Magazine latest issue, where he discusses how AI alone won’t solve pharma’s fragmented

Qinecsa Chief Product Officer, Beena Wood, has been featured in Pharma Time’s latest issue, where she reflects on the importance of groundwork before forging ahead

When Aurinia Pharmaceuticals launched their first FDA-approved product, LUPKNIS© (voclosporin), they recognised the need for a modern, scalable PV infrastructure. Reportum© was deployed which has
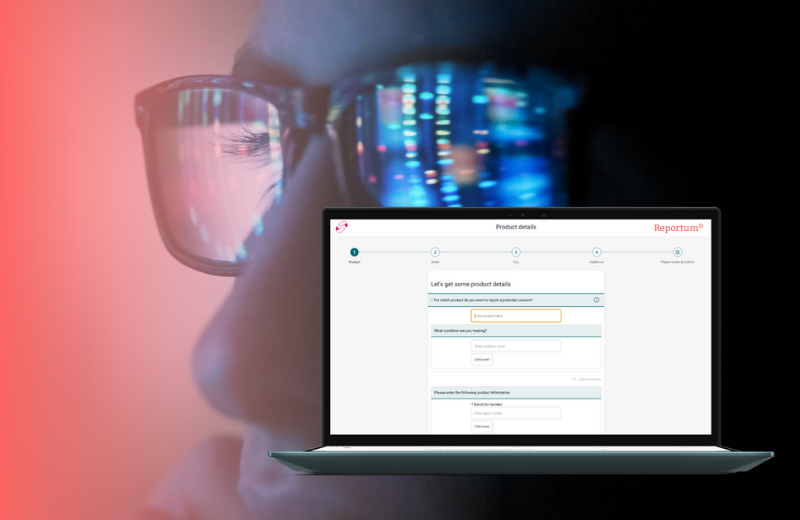
A global top-5 pharmaceutical company launched a groundbreaking therapy in one of it’s largest markets. Due to the success of the blockbuster drug, the team

Our client; a top-50 holistic healthcare company headquartered in Japan, globally markets several pharmaceuticals and nutraceuticals. Their challenge included processing cases from local affiliates worldwide

Qinecsa VP, Product Management QVW, Jon Messer, has been featured in European Pharmaceutical Contractor’s latest issue, where he discusses that using statistical methods in signal